Iranian Scientists Provide over 120 Million Doses of Acute Bird Flu Vaccine
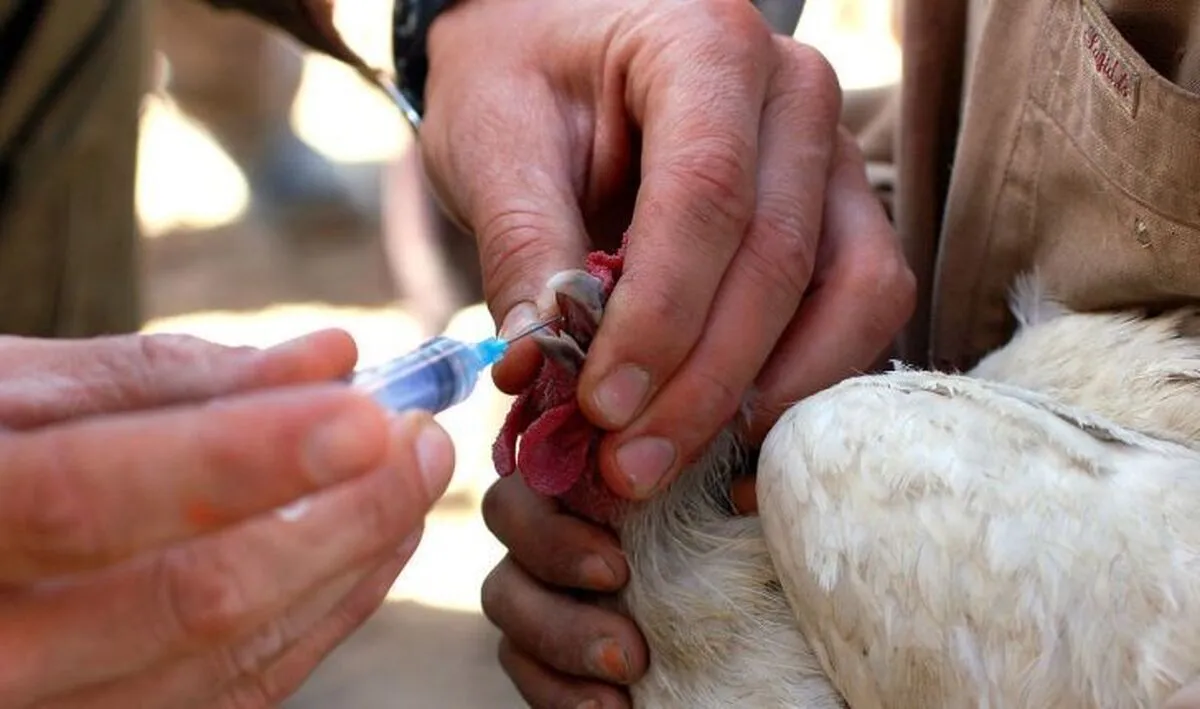
“Although no acute bird flu vaccine was imported to the country this year, a knowledge-based company which is a member of our association produced and distributed over 11 million doses of vaccine across the country,” said Abbas Jamali, a member of the Board of Directors of the Association of the Manufacturers and Exporters of Biotechnology Products of Iran.
He noted that since October 2020, over 120 million doses of the vaccine have been used in farms, egg-laying farms and turkey farms across the country, adding, “Fortunately, there have been no reports of complications or failure of the domestically-made vaccine to protect the herds so far.”
“No acute bird flu disease has been witnessed in these flocks,” Jamali said.
In a relevant development in October, Iranian researchers at the University of Tehran and Ferdowsi University of Mashhad for the first time in an inter-university collaboration succeeded in producing a recombinant H9N2 bird flu vaccine by using genetic engineering.
The success is the result of the specialized thesis of Amir Asqari, a student of the Faculty of Veterinary Medicine of the University of Tehran and it can reduce the symptoms of H9N2 bird flu and increase the level of immunity.
“To produce this vaccine, Pichia pastoris yeast was used which is a safe yeast, has probiotic properties, enjoys a very high ability to produce recombinant proteins and is free of endotoxin contamination,” said Jamshid Razmyar, an Associate Professor of the Faculty of Veterinary Medicine of the University of Tehran.
He added that information on how to make the vaccine has been submitted for patent and is in the final stages of judging, noting that the first article about the research was accepted in a Scopus-indexed journal (Iranian Journal of Veterinary Medicine) with the title ‘Serological Evaluation of H9-RBD-Pichia, a Novel Recombinant Influenza Vaccine, in BALB/c Mice’, and the second and third articles were also accepted.
Noting that the results of the research are now in the process of being submitted to the prestigious international journals, Razmyar said, “Necessary measures are being adopted to optimize the vaccine production in semi-industrial and then industrial conditions.”
4155/v





















